Mean kinetic temperature calculator excel
#Mean kinetic temperature calculator excel series#
(For recommended long-term testing conditions all over the world, see the WHO Technical Report Series No. They are an example of the worst-case scenario ideology often seen in the pharmaceutical and biotech industries. These temperatures are recommended by WHO in climatic zones I-II and (compared to the Haynes article) are probably high. In practice, products stored at controlled room temperature are often tested for long-term stability in simulated laboratory conditions of 25 or even 30 degrees (at 25☌ ± 2☌/60% RH ± 5% RH or 30☌ ± 2☌/65% RH ±5% RH) for dating purposes in climatic zones I-II, without using the exact calculated MKT value for this particular location. The description for stability testing conditions in countries located in Climatic Zones III (hot and dry) and IV (hot and humid) can be found in ICH Q1F explanatory note and in the WHO technical report “Annex 2: Stability testing of active pharmaceutical ingredients and finished pharmaceutical products.” Rules described in ICH Q1A (R2) are meant for climatic zones I-II (USA, EU and Japan).

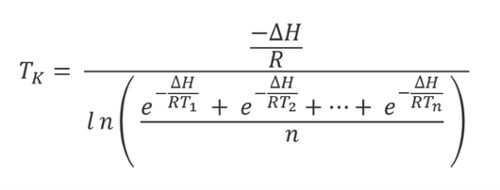
Grimm (1985, Drugs Made in West Germany). The WHO divides the world into four climatic zones: temperate, subtropical, hot/dry and hot/humid, based on the drug stability research presented by W. When establishing the temperatures for long-term stability testing of products to be stored at room temperature (RT) or controlled room temperature (CRT), the mean kinetic temperature in any part of the world can be derived from climatic data. It is based on the Arrhenius equation, which describes the temperature dependence of simple chemical reaction rates at ambient temperatures, where the rate of reaction generally doubles with every 10 degrees Celsius increase in temperature.

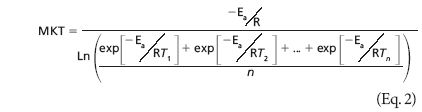
The equation he developed for “Virtual Temperature” is the same equation that is used today to calculate MKT. Haynes sought to address this variation by calculating a “Virtual Temperature” for use in expiry testing that would take into account the expected temperature variability in a given region. Simply put, changes in storage temperatures can affect the rate at which products degrade. The original purpose of the 1971 Haynes paper was to address the fact that climate-based temperature variation in uncontrolled pharmaceutical storage made it difficult to select a single temperature for use in product expiry testing. The document most commonly cited in GxP-regulated industries for the definition of mean kinetic temperature is the International Conference on Harmonization (ICH) guideline: “Stability Testing of New Drug Substances and Products Q1A (R2).” The MKT definition from this guideline is shown above. The math is difficult for most laypersons, and there is not a consensus on how MKT should be applied. MKT was first proposed to guide stability studies, and is now considered as a tool for evaluating temperature excursions in the dynamic arena of Good Distribution Practices. MKT can be a difficult tool to understand and apply properly. In the last 15 years of these ongoing efforts, mean kinetic temperature (MKT) has been identified as one of the potential tools available for evaluating the impact of temperature on product quality. Regulatory bodies and stakeholder organizations in drug and device manufacturing and distribution have long been working toward creating standards for temperature monitoring that ensure the shelf life, quality and safety of products.
